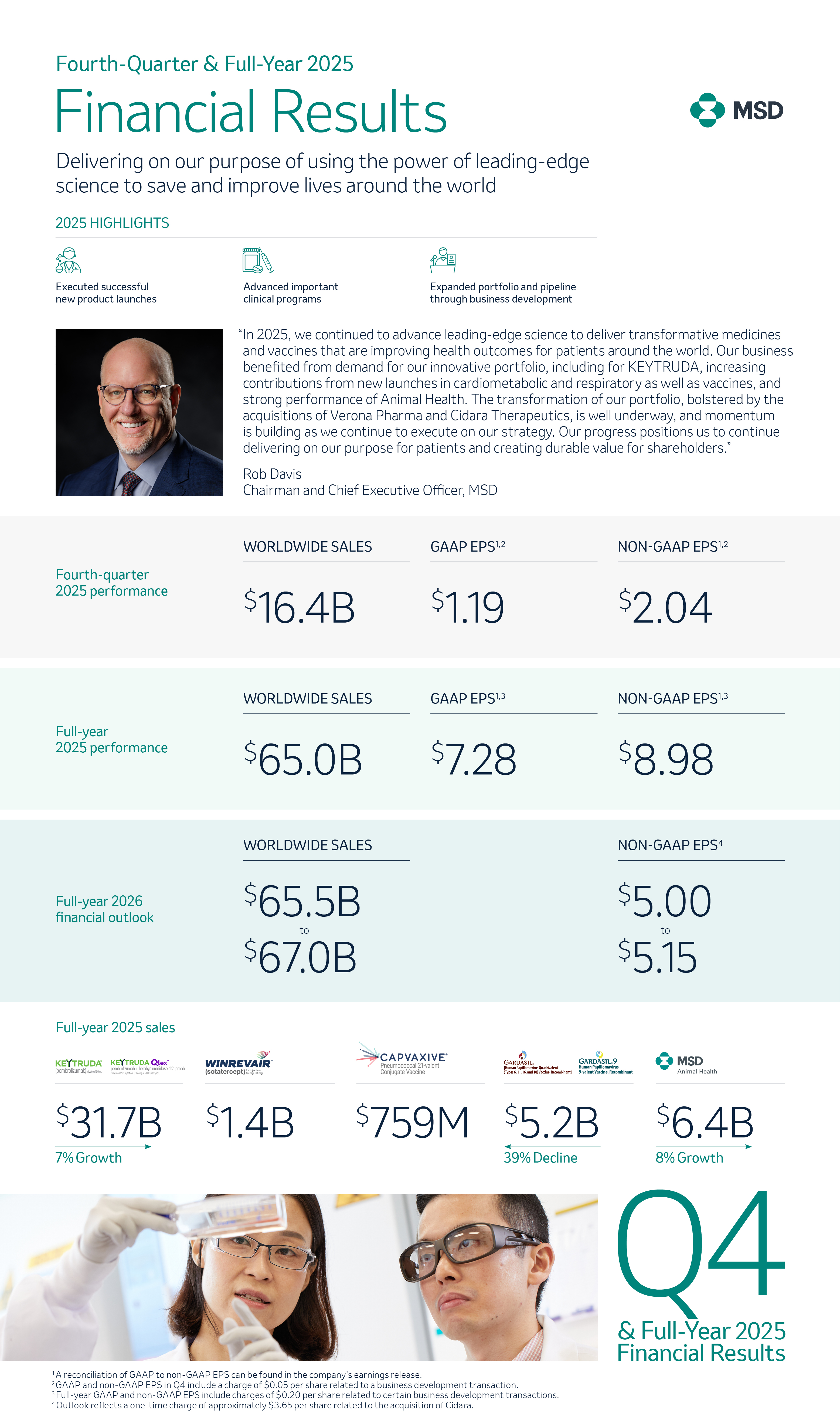
Expert Q&A: The role of real-world evidence in lung cancer detection
Shuvayu Sen, Ph.D., shared how our real-world evidence research uses data to analyze patient journeys and risk prediction models for early lung cancer detection
March 23, 2026

In the fight against lung cancer, early detection can be critical. While some countries have previously rolled out national lung cancer screening programs, in many cases, participation was low. This reality is one of the drivers behind Mission Lung Cancer, our collective effort to break down the barriers that stand in the way of early detection of lung cancer.
At the heart of our commitment to early lung cancer detection is one of our powerful contributions: scientific insights. Our real-world evidence (RWE) research uses patient-level data to analyze patient journeys and risk prediction models. This enables us to better understand diagnostic pathways and identify opportunities that may help facilitate early detection and diagnosis of lung cancer.
We spoke with Shuvayu Sen, Ph.D., vice president, oncology value and implementation outcomes research, about the importance of using RWE.
What is RWE and why does it matter?
Sen: RWE is generated through the analysis of real-world data or health information routinely collected from sources such as electronic health records (EHR), registries and insurance claims. Alongside data from clinical trials, real-world data matters because it may provide contextual insights that are not possible in a controlled setting.
How is your team using RWE in lung cancer research?
Sen: Our applicable areas of research include continuing to address smoking as the leading risk factor for lung cancer while identifying additional contributing risk factors — pinpointing moments to engage at-risk individuals and building explainable risk prediction models, including for non-small cell lung cancer. Our research on the patient journey can help show where delays in care may occur, such as low screening uptake, missed follow-ups on imaging or coordination gaps between care teams.
What have you learned from RWE in lung cancer?

Sen: As part of our analysis of one institution’s EHR database, we identified underutilization of low-dose computed tomography (CT) scans as an unmet need in the diagnostic pathway. Our research also showed potential for electronic medical record data to help identify patients who may be at risk of developing lung cancer. Looking ahead, we aim to explore options that could support earlier detection by leveraging this data. These insights point to potentially meaningful opportunities across the oncology ecosystem and beyond.
How else are we helping to advance research in this space?
Sen: We believe it’s critical to advance this work through research outside our company, as shared insights and investigator-led research are equally essential to accelerating innovation. That’s why we expanded our MSD Investigator Studies Program (MISP) to support independent research.
The MISP program evaluates tools and methods for lung cancer risk assessment and explores new technologies, like artificial intelligence (AI) and digital diagnostics, with the goal of improving early detection and diagnosis of lung cancer.
Together, our real-world evidence and MISP-supported research have the potential to reshape how and when lung cancer may be detected.
Learn more about Mission Lung Cancer.